 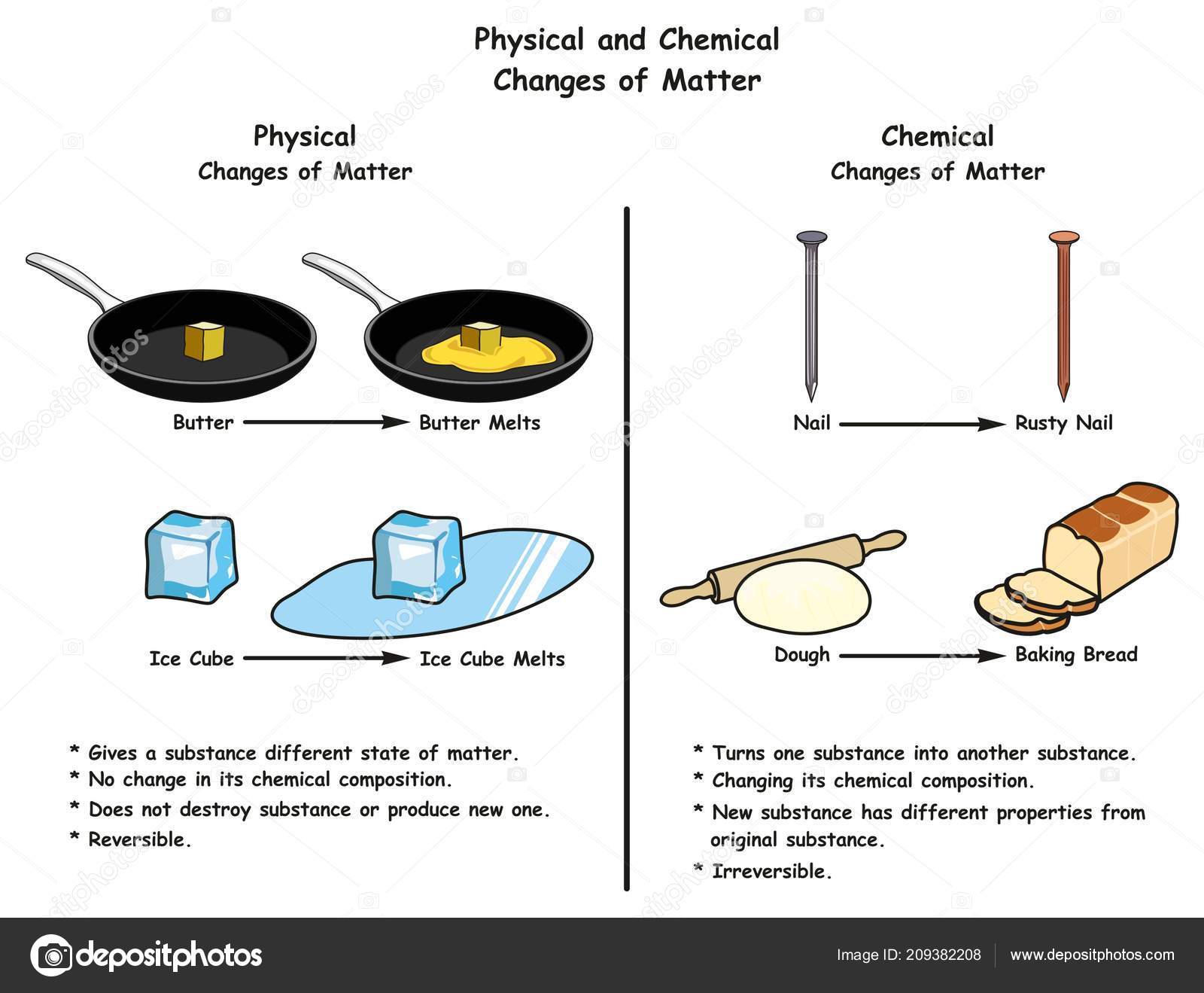
Now, what about physical properties like a boiling point? Boiling is something liquid can do, right? Not quite, by "can do", we mean "can do a chemical change". For example, Iron (Fe) can oxidize, but once it does, it's no longer iron! It is instead iron oxide (Fe(OH) 3). Once a substance actually does any of that, its identity (chemical composition) changes. Chemical properties are all about potential: the potential to catch fire, corrode, oxidize, etc. This can be a little confusing, so let's break it down. For example, flammability determines how likely something will catch fire.Ī key difference between a chemical and physical property is that a physical property describes what a substance currently is while a chemical property describes what a substance can do. It describes how it would behave in certain scenarios. Based on our chart, we see that ball 1 is aluminum, ball 2 is iron, and ball 3 is silver.Ī chemical property is the potential of a substance to undergo a chemical change. After measuring the density of each ball three times for the measure of accuracy, we get these results: ball 1 (2.72 g/cm 3), ball 2 (7.87 g/cm 3), and ball 3 (10.3 g/cm 3). Metal NameĪll of these metals are similar in color, but as you can see, their densities vary enough that we can tell them apart. Now, how can we tell them apart? One way is to determine their densities. They are all the same size and shape, and all of them are silver-colored. Let's say you were handed three metal balls. We can use these properties to determine the identity of unknown compounds or elements. Other examples of physical properties are malleability (the ability of a substance to be molded into thin sheets), hardness, solubility, and electrical conductivity. Intensive physical propertiesdo not depend on the amount of material, such as boiling point, density, or temperature.Įxtensive physical properties do depend on the amount, such as mass, length, and shape. So what are these properties exactly? Physical Properties A substance can have two main types of properties: chemical and physical. Everyone has their unique properties that help us identify them. Elements and chemical compounds work the same way. How would you find them? You can identify them by things like height, hair color, and how talkative they are. Let's say you had to pick your friend out of a crowd. What are physical and chemical properties? Lastly, we'll learn the key differences between these two.We'll identify examples of each of these changes.Then we will dive into the definition of physical and chemical changes.We will look at examples of these properties and how we can utilize them to identify unknown substances.Firstly, we will define physical and chemical properties.This article is about physical and chemical changes.In this article, we will learn what these changes, what they look like, and how they help us identify substances It can go through either a physical or chemical change. The Law of matter conservation states that matter cannot be created or destroyed, only changes its form.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
